When my friends and relatives ask me about the topic of my research, I first mention that I’m working in a research group specializing in the preclinical study of Alzheimer’s disease. Most of them have heard of Alzheimer’s disease or are affected by it, and I often face the same questions: How can I prevent from getting it myself? Why are so many elderly affected by it? Is there a cure against Alzheimer’s disease?
With a slight unease I usually provide an answer listing the factors that either lower or increase the risk of developing Alzheimer’s, explain the basic view of the disease progression and finally end my answer with a rather blunt but honest statement that there is currently no cure. This realization often leaves my audience, myself included, somewhat deflated. But why is it that over hundred years after the first description of the disease by doctor Alois Alzheimer and thousands of subsequent studies there still isn’t a disease-modifying treatment? Maybe a small review is in order.
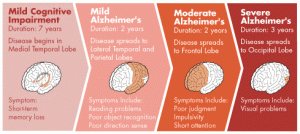
Alzheimer’s disease is the most common cause of dementia in the elderly. Age is the primary risk factor for Alzheimer’s disease. The risk of developing the disease doubles every five years after 65 years of age, being roughly 5% at 65 years and 40% at 80 years. The disease is thought to start from near the medial temporal lobe, where the memory center, hippocampus, is located, and then spread outwards to the perimeter of the brain, the cortex (see Picture 1). As a result, the first detectable behavioral symptoms of the disease are often memory related problems. Death of neurons, the brain cells responsible for receiving, processing and transmitting signals needed for our thought processes and bodily functions, and the loss of the connections between them correlates with the spread of the pathology and symptoms.
Picture 1: The progression of Alzheimer’s disease pathology and symptoms
(www.preventad.com/ad_symptom.html)
And what causes this progressive brain damage? Currently there are only mostly educated guesses on when and what exactly starts the pathology, but the main culprits behind the pathology are thought to be the proteins amyloid-beta (Ab) and Tau. Ab is a fragment of the amyloid precursor protein (APP) and is normally cleared from the brain, but accumulates in Alzheimer’s disease to form deposits called amyloid plaques (not to be confused with plaque formation in the teeth!). Before forming plaques, Ab disrupts neuronal signaling and causes toxicity. Tau on the other hand is a structural component of neurons that normally stabilizes microtubules that form part of the cell’s backbone called the cytoskeleton. In Alzheimer’s disease Tau is modified by enzymes called kinases to dissociate from microtubules, forming disruptive tangles leading to further neuronal toxicity and dysfunction. Ultimately the affected neurons die due to the accumulation of stress and damage. Familial mutations in Ab, Tau and associated genes are known to cause an aggressive Alzheimer’s pathology and an earlier onset of the disease (before 65 years).
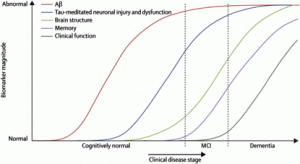
Here comes the major dilemma of Alzheimer’s disease: deposition of the aforementioned plaques and tangles begins years, even decades, before the onset of the first detectable symptoms (see Picture 2). At this stage over 50% of brain cells in specific regions might already be dead or beyond rescue. The regeneration of brain cells, especially of neurons, is very limited, but the brain has an enormous adaptive capability to cope with damage by rewiring. This can efficiently mask the pathological onset of Alzheimer’s disease that may precede the first symptoms by even 20 years! MCI, or mild cognitive impairment, is considered a stepping-stone to Alzheimer’s, but not everyone with MCI will develop the disease since memory also suffers during the normal aging process. This presents the first major hurdle in curing Alzheimer’s disease: how to find the future Alzheimer’s patients before any visible symptoms or widespread pathology has developed?
Picture 2: Progression of the pathology and biomarker magnitude in Alzheimer’s disease (Jack CR Jr, Knopman DS, Jagust WJ, et al, 2010 Lancet)
Luckily, the search for clues or biomarkers of early Alzheimer’s pathology is well underway. Techniques to detect Ab and Tau from blood, CSF (cerebrospinal fluid) and by brain imaging are developing fast, but they are costly and the reliability of pre-diagnosis is still low. Some nationwide initiatives have been taken to find reliable markers to detect early pathological biomarkers from blood. One such endeavour is the Australian Imaging, Biomarker and Lifestyle (AIBL) study that has yielded valuable insight into the correlation between Ab accumulation in the brain and biomarkers in blood. Such studies can potentially provide an inexpensive and accurate diagnostic tool to assess Alzheimer’s risk in the future.
The second major challenge remains to find a disease-modifying treatment. Even the most recent clinical trials aiming to reduce Ab with an antibody have failed to produce a significant effect in patients suffering from mild-to-moderate Alzheimer’s disease. However, earlier treatment might offer improved efficacy when targeted at the early stages of Ab and Tau pathology. The conclusions from these studies are often uniform: Targeting or preventing the early pathological changes holds the best chance for an efficient disease-modifying treatment.
But until potent preventive treatments are developed, it is safest to follow the common formula for a healthy lifestyle: eat your nutrient-rich vegetables, pick the omega-3 rich fatty fish over the red meat, steer clear of excessive drinking and keep socially and physically active. To further decrease your risk of Alzheimer’s, educate yourself and challenge your brain with crosswords and Sudoku, even on holidays. And if you’re worried about your parents or grandparents, then simply make more time to see them. Perhaps treat them to a nice meal with salmon and a good bottle of resveratrol-rich New Zealand Pinot Noir, or maybe challenge them in a weekly round of golf. For those moments are surely ones that are hard to forget.
About Me:
Kai Kysenius, M.Sc., PhD student
Neuroscience Center, University of Helsinki
www.mv.helsinki.fi/home/hjhuttun/

Witty Article, I very much enjoyed reading it! I must agree with your last statement; resveratrol-rich Pinot Noir is indeed something one should indulge in more often! Good for your heart, and good for your brain. I like!