Niskakoski A, Pasanen A, Porkka N, Eldfors S, Lassus H, Renkonen-Sinisalo L, Kaur S, Mecklin JP, Bützow R, Peltomäki P.
OBJECTIVE: The diagnosis of carcinoma in both the uterus and the ovary simultaneously is not uncommon and raises the question of synchronous primaries vs. metastatic disease. Targeted sequencing of sporadic synchronous endometrial and ovarian carcinomas has shown that such tumors are clonally related and thus represent metastatic disease from one site to the other. Our purpose was to investigate whether or not the same applies to Lynch syndrome (LS), in which synchronous cancers of the gynecological tract are twice as frequent as in sporadic cases, reflecting inherited defects in DNA mismatch repair (MMR).
METHODS: MMR gene mutation carriers with endometrial or ovarian carcinoma or endometrial hyperplasia were identified from a nationwide registry. Endometrial (n = 35) and ovarian carcinomas (n = 23), including 13 synchronous carcinoma pairs, were collected as well as endometrial hyperplasias (n = 56) and normal endometria (n = 99) from a surveillance program over two decades. All samples were studied for MMR status, ARID1A and L1CAM protein expression and tumor suppressor gene promoter methylation, and synchronous carcinomas additionally for somatic mutation profiles of 578 cancer-relevant genes.
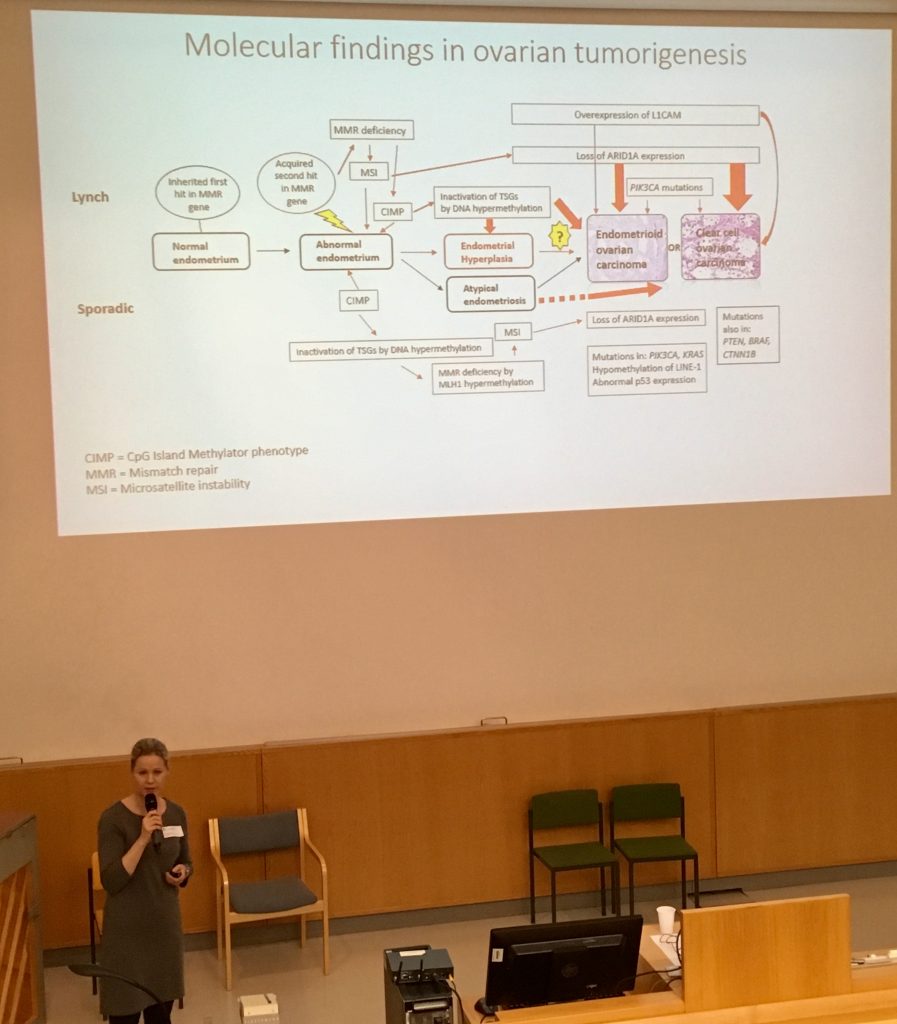
RESULTS: Synchronous carcinomas were molecularly concordant in all cases. Prior or concurrent complex (but not simple) endometrial hyperplasias showed a high degree of concordance with endometrial or ovarian carcinoma as the endpoint lesion.
CONCLUSIONS: Our investigation suggests shared origins for synchronous endometrial and ovarian carcinomas in LS, in analogy to sporadic cases. The similar degrees of concordance between complex hyperplasias and endometrial vs. ovarian carcinoma highlight converging pathways for endometrial and ovarian tumorigenesis overall.
Gynecol Oncol. 2018 Apr 28. doi: 10.1016/j.ygyno.2018.04.566. [Epub ahead of print]
PMID:29716739