by Arto Heitto, Liine Heikkinen, Nina Sarnela, Otso Peräkylä, Yuqin Liu and Heikki Junninen
Arctic areas are warming fast due to global warming, and one of the most striking effects of this warming is the rapid shrinking of sea ice. Records for the lowest sea ice are being broken nearly annually. Just this spring we saw another record low winter sea ice extent (announcement from NASA). The diminishing sea ice cover has many effects. Probably the most well-known of these is related to ice being bright, but open sea being dark in colour. This means that sea ice reflects the majority of sunlight back to space, while open water absorbs most of it. So when ice cover decreases, more of the sunlight falling on arctic areas is absorbed, leading to warming of the surface (check this video: https://svs.gsfc.nasa.gov/20021). This warming in turn leads to more sea ice loss: this is a type of mechanism called a positive feedback. In such a mechanism, an initial change to a system (in this case warming of the Arctic), leads to accelerated, further changes (more warming as more radiation is absorbed by the surfaces).
However, changing brightness of the surface is not the only change caused by diminishing sea ice. On top of the change of brightness of the surface, there are also many other differences between water and ice covered surfaces. Ice acts as a lid between the water and the air. In ice covered regions, matter (like water vapour, carbon dioxide or other gases) cannot easily move from the water to the air and vice versa. We say that the exchange between the sea and the atmosphere is reduced. However, sea ice can provide different methods for transport of material to the atmosphere. For example, when sea ice is formed, pure water freezes first. This leaves miniature pockets of very salty water, or brine, still in liquid form. Different components of the sea water can then possibly escape to the atmosphere more easily from this concentrated solution.
So why would we be interested in the transport of stuff from the sea to the atmosphere? There are many reasons. Sea water is rich in all kinds of chemicals, which can have big impacts on the chemistry of the atmosphere, if they manage to escape the sea and get to the air. Examples of these kinds of compounds include sulfur containing compounds produced by different types of plankton, and sea salt itself. Roughly half of sea salt is composed of chloride ions. In their ionic, or electrically charged form, they are relatively harmless. However, in electrically neutral form, they become extremely reactive chemically. The same applies to other components of sea salt found in lesser quantities, including bromine and iodine. Though there is less of these compounds in the water, they are even more reactive than chlorine. Chlorine, bromine and iodine are all part of a group of compounds called halogens (see what others are planning to investigate: http://airbornescience.jpl.nasa.gov/campaign/bromex). If they manage to get into the atmosphere, they can have big impacts on how pollutants are processed in the air. Additionally, they can in some conditions form new tiny solid or liquid droplets, so called aerosol particles. To get an idea of their size, a human hair is roughly 100 micrometres thick on average. The size of aerosol particles varies from 1 nanometre, a thousandth of a micrometer, to roughly 10 micrometres. So the smallest aerosol particles are only one hundred thousandth of the diameter of human hair, though the biggest ones are already close this size. So what do these aerosol particles do? They have many impacts, ranging from health effects to visibility. The most important in this context is their effect on climate. Through reflecting sunlight back to space, or making clouds brighter and longer lived, they can cool the climate.
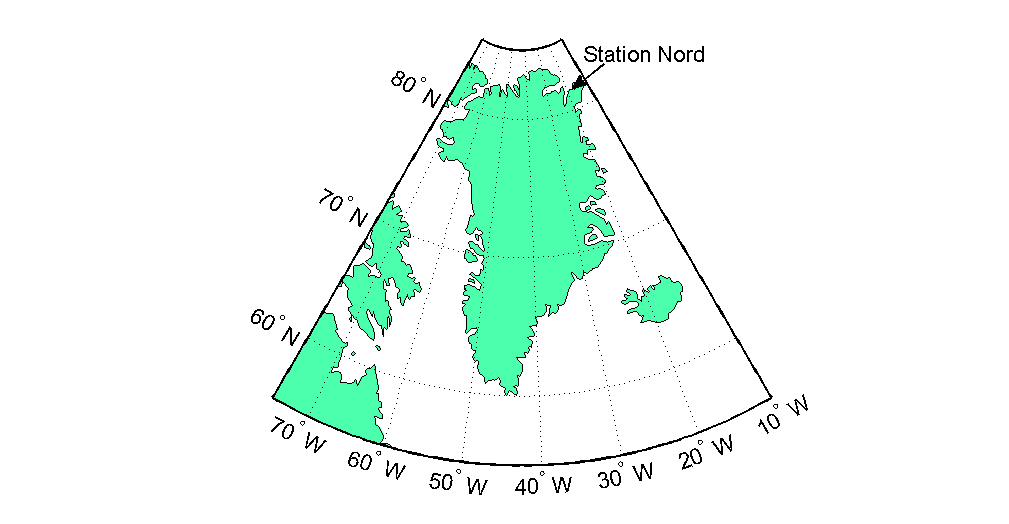
But back to sea ice. So ice-covered and ice-free surfaces can differ in how well they allow chemicals to pass from the sea to the atmosphere. These chemicals can in turn lead to formation of aerosol particles that can cool the climate. Thus, when sea ice cover is reduced, it also affects this process of aerosol formation. In order to figure out how exactly the process is affected, we analyzed data measured in the Villum Research Station, Northeastern Greenland, located next to the Danish military station, Station Nord. We measured various parameters in the station, including halogens in their oxidized form, different sulfur-containing species, ozone, and aerosol particles of various sizes, among others. To figure out where these compounds came from, we compared the measured concentrations to where the air had come from.
One of our findings was that sea ice seemed to be a source of aerosol particles: the more time the air had spent over ice, the more aerosols it had. Also, it seemed that air passing over open water sometimes had higher concentrations of some halogens: this could mean that in the future we can get even more of those. In total, we found many potentially interesting links between sea ice and air chemistry in the Arctic. However, all of these require further study before reliable conclusions can be made about potential future impacts.




 Almost every day we hear about how bad climate change is for the Earth and the population. Yet after speaking with locals in Nuuk and experiencing a small part of the Greenlandic culture, we got some other perspectives. In Greenland, a vast island almost wholly covered by the huge ice sheet, many of the residents view the climate change in a positive light.
Almost every day we hear about how bad climate change is for the Earth and the population. Yet after speaking with locals in Nuuk and experiencing a small part of the Greenlandic culture, we got some other perspectives. In Greenland, a vast island almost wholly covered by the huge ice sheet, many of the residents view the climate change in a positive light.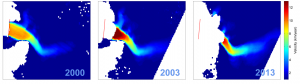
 Scientists love graphs and charts and read them as a second language, but the complicated pictures you find in scientific journals can look like gibberish for the general public. One of our group’s task during the #ArcticCourse was to find new ways to visualize climate and environmental data from Arctic. We brainstormed what kind of data would give new insights and how to present it in modern way. Here we tell about some of these ideas.
Scientists love graphs and charts and read them as a second language, but the complicated pictures you find in scientific journals can look like gibberish for the general public. One of our group’s task during the #ArcticCourse was to find new ways to visualize climate and environmental data from Arctic. We brainstormed what kind of data would give new insights and how to present it in modern way. Here we tell about some of these ideas. 





