New Drugs for Heart Diseases
The human heart beats approximately 100,000 times every day. Pumping oxygen-rich blood across our body, the heart is essential in keeping our other organs healthy. A heart attack occurs when cardiac cells, including the beating heart muscle cells (cardiomyocytes) are themselves deprived of oxygen due to a blockage in a blood vessel in the heart. This in turn leads to the death of these cells which are incapable of regrowing, and therefore reduces the heart’s pumping capacity. This phenomenon is known as heart failure. Similarly, high blood pressure or genetic conditions can cause the heart muscle to stiffen or enlarge, decreasing the heart’s ability to pump blood.
We aim to address some of the fundamental questions pertaining to heart diseases. One of our goals is to identify ways to force remaining heart muscle cells to regrow (proliferate) to heal the heart after a heart attack. Stem cells are excellent tools for such studies as they can become any cell type of the human body. In our lab, we generate heart muscle cells and blood vessel endothelial cells from stem cells and study the effects of different conditions, drugs, or chemical compounds on these cells.
Learn more about Dr Qasim Majid and Dr Virpi Talman from the Regenerative Cardiac Pharmacology lab.


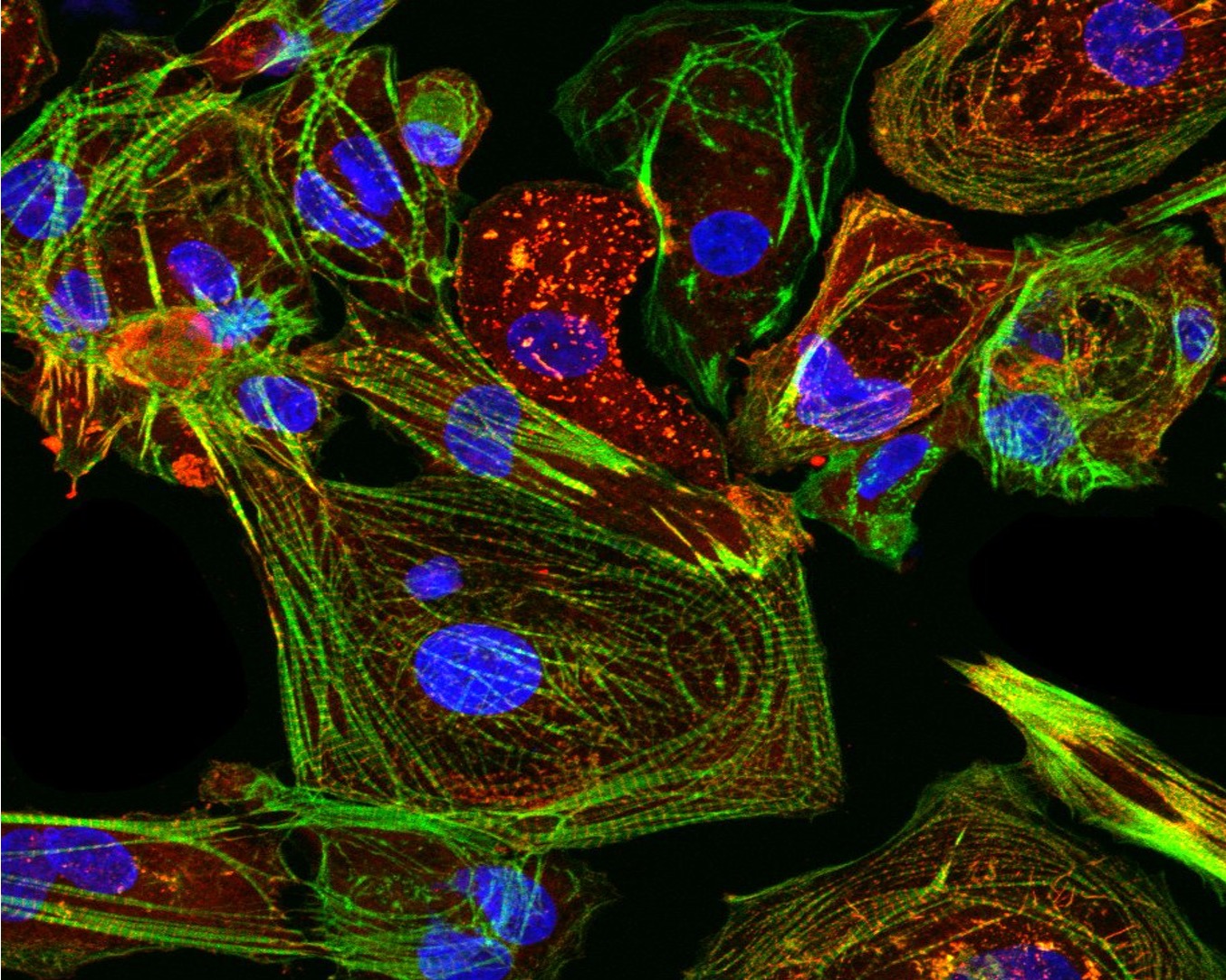
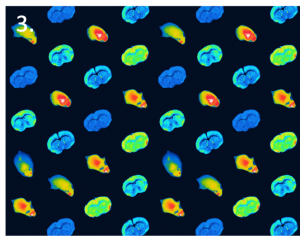
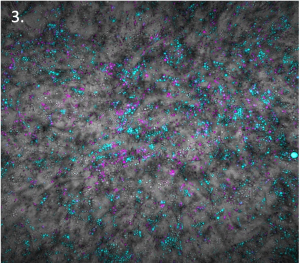
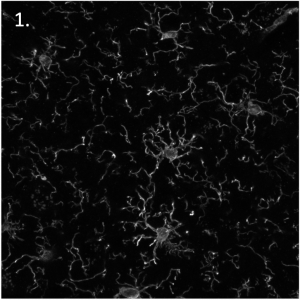
Fluorescence microscope images of:
- Human pluripotent stem cell-derived cardiomyocytes.
- Human pluripotent stem cell-derived cardiomyocytes and endothelial cells.